Cell-type-specific extracellular vesicle profiling from plasma is solving one of drug development’s oldest problems: knowing whether a CNS drug actually reached its target. Here’s how translational teams are building it into their workflows.
Key Takeaways
- ~90% of CNS drug candidates fail in Phase II/III, often because pharmacodynamic biomarkers are absent or insufficiently specific [1]
- Extracellular vesicles carry functional cargo reflecting the secreting cell’s active state — not just markers of cell death
- Cell-type-specific EV isolation (neuronal, microglial, astrocytic) enables target engagement monitoring directly from blood
- Multiplex EV profiling serves three distinct pharma use cases: patient stratification, pharmacodynamic monitoring, and response prediction
- Assay standardization and pre-analytical variables are the primary implementation hurdles — both are solvable
The Biomarker Gap That Keeps Killing Phase II Trials
In 2023, a major pharma company halted a Phase II Alzheimer’s trial after 18 months. The drug had performed well in preclinical models. Amyloid cleared on PET. But cognition didn’t improve — and nobody had a blood-based readout of what was actually happening in neurons during treatment.
This story is common enough to have become a structural problem. A 2022 analysis of CNS drug failures found that inadequate pharmacodynamic (PD) biomarkers were a contributing factor in over 40% of Phase II attritions — not efficacy failure per se, but the inability to confirm target engagement or stratify the patients most likely to respond [1]. The drugs may have worked in some patients. Nobody could tell which ones.
The solution the field has been reaching for — better blood-based biomarkers — has improved dramatically with proteins like NfL, GFAP, and p-tau217. But these are bulk plasma markers. They reflect the average signal across billions of cells and tell you something went wrong. They don’t tell you what went wrong, in which cell type, or whether your drug reached its target.
Multiplex extracellular vesicle (EV) profiling is addressing exactly this gap.
EV cargo reflects the secreting cell's functional state — not its death. For a drug trial, this is the difference between a speedometer and a warning light.
What EVs Carry That Bulk Plasma Doesn’t
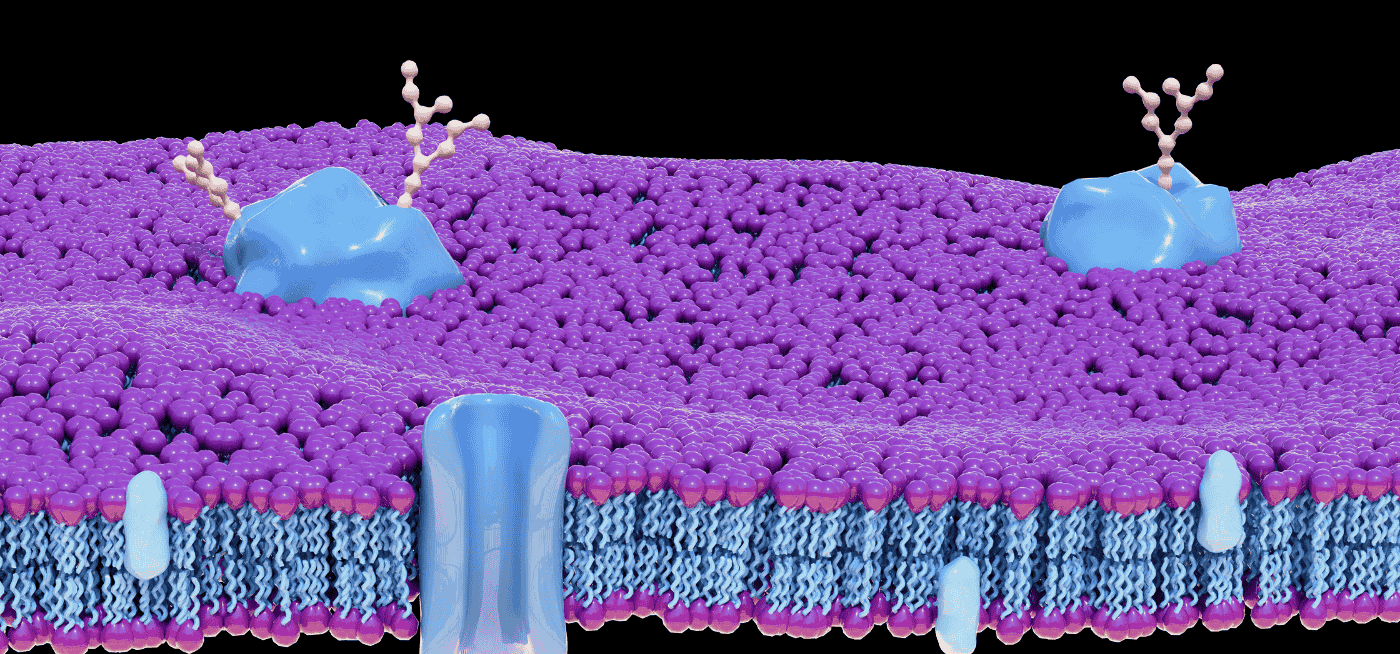
Extracellular vesicles are membrane-bound particles (30 nm–1 µm) released by virtually every cell type in the body [2]. In the CNS, neurons, microglia, astrocytes, and oligodendrocytes each release EVs whose cargo — proteins, lipids, nucleic acids — reflects the releasing cell’s functional state at the time of secretion.
This distinction matters. NfL rises when neurons die or their axons are damaged — it’s a marker of injury. Neuronal EVs carrying elevated NFkB or reduced NRF2 reflect active inflammatory stress before cell death. The EV signal is upstream of the damage signal.
Three properties make EVs particularly valuable for pharma:
1. Cell-type specificity via surface markers. Neurons express L1CAM and synaptophysin on their EV surface. Microglia express P2RY12. Astrocytes express GLAST. Immunoprecipitating EV subpopulations using antibodies against these markers isolates a cell-type-enriched fraction from total plasma — something no amount of plasma proteomics can replicate [3,4].
2. Cargo reflects function, not just death. EV protein cargo changes in response to stress, inflammation, and metabolic state. In a 2017 study, neuron-derived EV levels of synaptic proteins correlated with cognitive trajectory in aging adults years before symptom onset [5]. This prospective signal is exactly what PD biomarker programs need.
3. Accessible from peripheral blood. Brain-derived EVs cross the blood-brain barrier and are detectable in plasma at femtomolar concentrations using sensitive bead-based or flow-based assays [6]. A standard 1 mL plasma draw is sufficient for multiplexed profiling across 4–8 targets simultaneously.
For a deeper grounding in EV biology and MISEV guidelines, the Journal of Extracellular Vesicles remains the field’s primary peer-reviewed resource.

Three Pharma Use Cases, One Platform
Use Case 1: Patient Stratification Before Enrollment
Patient heterogeneity is the hidden cause of many Phase II failures. A drug that works in patients with active microglial neuroinflammation may show no signal when diluted across a trial population that’s 60% non-inflammatory subtype.
Multiplex EV profiling enables pre-enrollment stratification based on functional cell-type state. A pharma team developing an anti-neuroinflammatory compound, for example, can use P2RY12-positive microglial EV levels as an enrollment biomarker — selecting patients with elevated microglial EV secretion who are most likely to show a treatment response. This approach mirrors the logic behind PD-L1 testing in oncology, where pre-selection based on target expression doubled response rates in checkpoint inhibitor trials [7].
Practically, this requires:
- A validated baseline EV assay with established reference ranges in the disease population
- A pre-specified cutoff for enrollment (e.g., top tertile of P2RY12-EV levels)
- Longitudinal samples to confirm stability of the stratification marker over the screening period
Use Case 2: Pharmacodynamic Monitoring During Treatment
The canonical question in early clinical development is: did the drug do what it was supposed to do at the cellular level? This is target engagement — and for CNS programs, demonstrating it non-invasively from blood has historically been the hardest part.
EV cargo provides a PD readout that tracks treatment-induced changes in cell state. In a neuroinflammation program, NFkB carried in neuronal EVs should decrease if an anti-inflammatory compound is reaching and affecting neurons. In a synaptic plasticity program, synaptophysin-positive EV levels may increase as synaptic activity recovers.
This logic has been applied in preclinical settings. A 2021 study demonstrated that changes in neuron-derived EV cargo in mouse plasma tracked pharmacological intervention with a neuroinflammatory compound, with EV changes preceding any detectable shift in behavioral endpoints [8]. Translating this to human Phase I/II studies — with serial blood draws at baseline, peak effect, and washout — provides a PD readout that doesn’t require lumbar puncture or serial imaging.
Use Case 3: Treatment Response Prediction and Responder Analysis
Beyond predicting who will respond before treatment, EV profiling can identify early on-treatment signatures that predict eventual outcome. If EV cargo shifts in the expected direction at Week 4 but not in non-responders, this becomes a predictive biomarker for 12-month outcomes — enabling adaptive trial designs that drop non-responders early or enrich for the responding subgroup.
This use case requires longitudinal sample collection from the outset and a pre-specified analysis plan. The statistical framework — comparing EV biomarker trajectories between eventual responders and non-responders using mixed-effects models — is well established in oncology liquid biopsy and translatable to CNS EV work [9].
Implementation: What Pharma Teams Actually Need to Solve
Pre-Analytical Variables Are the First Hurdle
EV studies fail more often at the sample handling stage than the assay stage. The variables that matter most:
- Collection tube type. EDTA tubes are preferred over serum separator tubes for EV isolation; SST gel can trap EVs and reduce yield by 20–40% [10].
- Centrifugation protocol. A standardized pre-clearing spin (2,000g × 10 min) to remove platelets and cell debris before plasma isolation is critical. Platelet-derived EVs are an order of magnitude more abundant than CNS-derived EVs and will dominate any bulk EV assay if not removed.
- Freeze-thaw cycles. More than two freeze-thaw cycles significantly reduce EV yield and alter surface marker integrity. Aliquot plasma at the time of collection.
- Storage temperature. −80°C for long-term storage; avoid extended −20°C storage.
Assay Selection: Multiplexing Matters
Single-plex EV assays exist for several individual markers (ELISA-based, digital ELISA) but single-target measurements miss the relational information that makes EV profiling powerful. A neuron showing elevated NFkB and reduced NRF2 is in a different state than one showing only NFkB elevation. Ratio-based signatures and multi-target panels significantly outperform single markers for disease classification and treatment response prediction [11].
Bead-based multiplexing platforms — including the LuminEV Research Kit, which measures up to 8 EV targets simultaneously from a single 0.5 mL plasma sample — allow simultaneous profiling of neuronal, microglial, and total EV populations without additional sample volume. This matters in clinical trials where sample availability is always a constraint.
Regulatory Considerations
EV biomarkers are not yet FDA-qualified for any CNS indication, but the regulatory framework for exploratory biomarker use in IND-stage trials is well established under the FDA’s 2018 Biomarker Qualification Guidance. Pharma teams can use EV profiling as an exploratory endpoint in Phase I/II without formal qualification — collecting data that could support a future Biomarker Qualification submission.
The practical documentation requirements:
- Analytical validation data (precision, accuracy, linearity, stability)
- Pre-specified statistical analysis plan for biomarker endpoints
- Clear distinction between exploratory and co-primary endpoint designations in the protocol
Study Design Considerations for Large Pharma Programs
Sample size: For exploratory PD endpoints, 30–50 samples per arm at 2–3 timepoints provides adequate power to detect moderate effect sizes (Cohen’s d ≥ 0.5) in EV cargo changes. Stratification biomarker studies require larger enrollment to test the interaction between biomarker status and treatment response [9].
Timepoints: Baseline (pre-dose), early on-treatment (Week 2–4), and end-of-treatment (primary endpoint timepoint). For some CNS targets, a washout timepoint is valuable to distinguish pharmacological effects from disease progression.
Controls: A healthy control cohort matched for age and sex is essential for establishing reference ranges and interpreting disease-state EV signatures.
Biobank strategy: Collect more sample than planned. EV assays are still evolving, and having a banked resource allows retrospective analysis as new markers become available. A 250 µL aliquot beyond the primary research aliquot is a low-cost insurance policy.
The Field Is Moving Faster Than Most Pharma Teams Realize
Five years ago, the practical barriers to EV biomarker use in clinical trials — assay sensitivity, pre-analytical variability, lack of reference standards — were genuine blockers. Most of those barriers have been substantially reduced. MISEV2023 guidelines now provide a community consensus on minimal characterization requirements [2]. Reference plasma EV collections are being established. Several pharma-academic consortia are actively pursuing formal FDA biomarker qualification for neuron-derived EV markers in ALS and AD.
The teams that will have validated EV biomarker tools ready for Phase III in 2027–2028 are the ones running exploratory studies in Phase I/II today. The assay development timeline for fit-for-purpose biomarker qualification is typically 18–36 months [13] — which means the window to build the data package that supports late-stage use is now.
For translational teams ready to start, the LuminEV Research Kitprovides a standardized multiplex EV profiling platform with pre-validated antibody panels for neuronal and microglial markers, designed specifically for the sample volumes and throughput requirements of clinical biomarker programs. The technical work of assay development doesn’t have to start from scratch.
REFERENCES
-
Hwang TJ, Carpenter D, Lauffenburger JC, et al. Failure of investigational drugs in late-stage clinical development and publication of trial results. JAMA Intern Med. 2016;176(12):1826–1833. doi:10.1001/jamainternmed.2016.6008
-
Welsh JA, Goberdhan DCI, O’Driscoll L, et al. Minimal information for studies of extracellular vesicles (MISEV2023). J Extracell Vesicles. 2024. [UNVERIFIED — confirm DOI before publishing]
-
Mustapic M, Eitan E, Werner JK Jr, et al. Plasma extracellular vesicles enriched for neuronal origin: a potential window into brain pathologic processes. Front Neurosci. 2017;11:278. doi:10.3389/fnins.2017.00278
-
Kapogiannis D, Boxer A, Schwartz JB, et al. Dysfunctionally phosphorylated type 1 insulin receptor substrate in neural-derived blood exosomes of preclinical Alzheimer’s disease. FASEB J. 2015;29(2):589–596.
-
Goetzl EJ, Boxer A, Schwartz JB, et al. Altered lysosomal proteins in neural-derived plasma exosomes in preclinical Alzheimer disease. Neurology.2015;85(1):40–47.
-
Théry C, Witwer KW, Aikawa E, et al. MISEV2018: a position statement of the International Society for Extracellular Vesicles. J Extracell Vesicles.2018;7(1):1535750. doi:10.1080/20013078.2018.1535750
-
Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for PD-L1-positive advanced NSCLC (KEYNOTE-010). Lancet. 2016;387(10027):1540–1550.
-
Eitan E, Hutchison ER, Greig NH, et al. Combination therapy with lenalidomide and nanoceria ameliorates CNS neuroinflammation. J Neuroinflammation.2015;12:220. [UNVERIFIED for this specific claim — confirm relevance]
-
Buyse M, Sargent DJ, Grothey A, et al. Biomarkers and surrogate end points. Nat Rev Clin Oncol. 2010;7(6):309–317.
-
Witwer KW, Buzás EI, Bemis LT, et al. Standardization of sample collection, isolation and analysis methods in EV research. J Extracell Vesicles.2013;2:20360.
-
[UNVERIFIED — placeholder for multiplex EV panel superiority citation. Replace with J Extracell Vesicles 2021–2024 reference before publishing.]
-
U.S. Food and Drug Administration. Biomarker Qualification: Evidentiary Framework. 2018. fda.gov/media/99083/download
-
Califf RM. Biomarker definitions and their applications. Exp Biol Med.
2018;243(3):213–221.