A deep dive into extracellular vesicle surface proteomics, intercellular signaling mechanisms, and their transformative role in diagnostics and therapeutics.
Key Topics: Extracellular vesicles · EV surface proteins · Cell communication · Exosome proteomics · EV biomarkers · Intercellular signaling · Liquid biopsy
Introduction: The Hidden Language of Cells
Every cell in the human body is engaged in constant conversation. Far from operating in isolation, cells exchange molecular messages that coordinate tissue development, regulate immune responses, and maintain physiological homeostasis. For decades, scientists assumed this communication relied primarily on soluble factors — hormones, cytokines, and growth factors — diffusing through extracellular fluids. That assumption has been fundamentally revised.
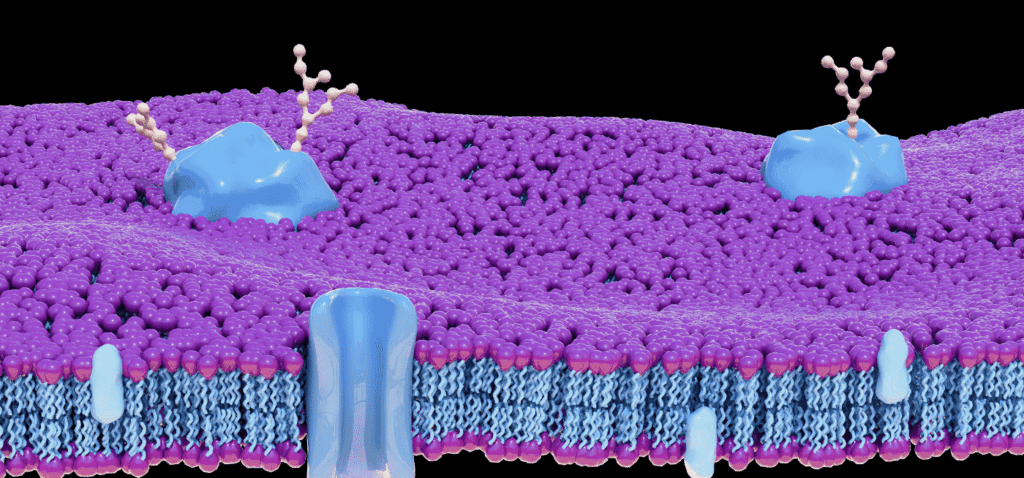
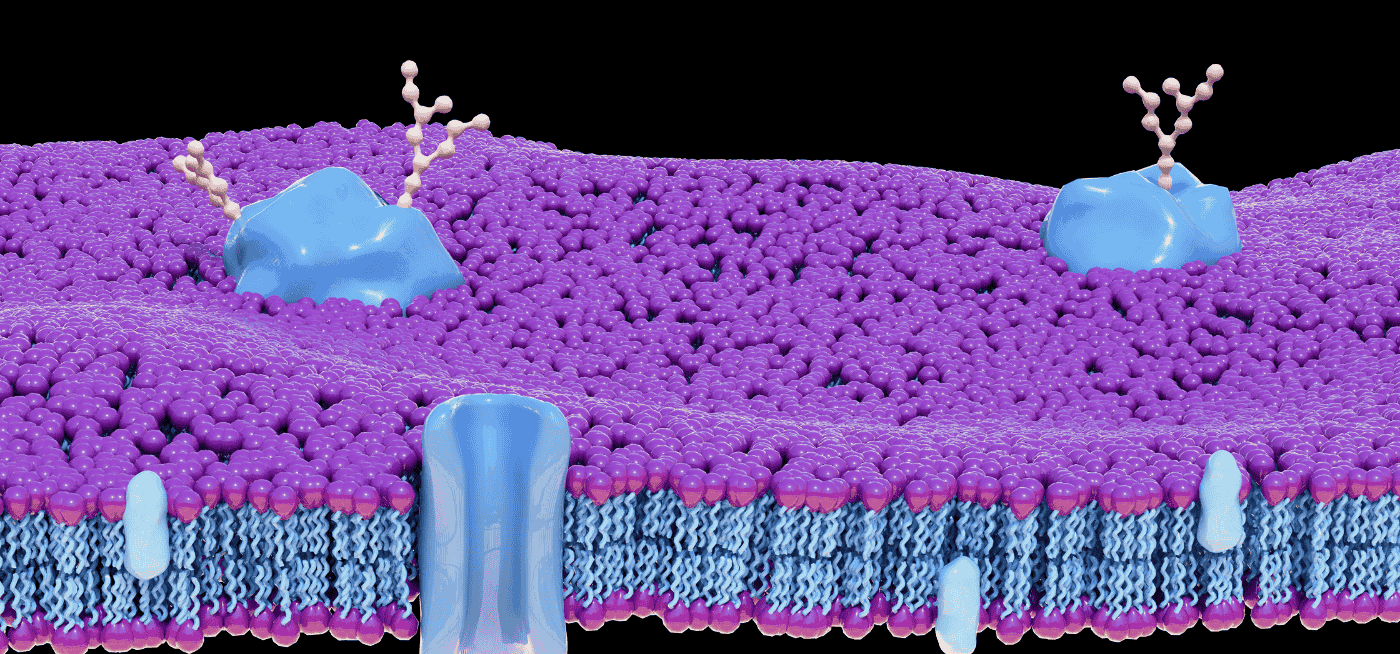
We now understand that cells release a diverse population of membrane-enclosed particles called extracellular vesicles (EVs), which serve as sophisticated couriers of biological information. These nanoscale vehicles carry cargo ranging from nucleic acids and lipids to metabolites and, crucially, a complex repertoire of surface-displayed proteins. It is these EV surface proteins that function as both the address label and the key — determining where a vesicle travels and how it interacts with recipient cells.
At LuminEV, our research centers on decoding this molecular language. Understanding the surface proteome of extracellular vesicles opens extraordinary windows into how cells communicate in health and disease — and offers transformative opportunities in diagnostics, drug delivery, and therapeutic intervention.
What Are Extracellular Vesicles?
Extracellular vesicles are a heterogeneous population of lipid bilayer-enclosed structures secreted by virtually all cell types. They are broadly classified into three main subtypes based on their biogenesis, size, and surface markers:
- Exosomes (30–150 nm): Formed through the endosomal pathway when multivesicular bodies (MVBs) fuse with the plasma membrane. Exosomes are enriched in tetraspanins such as CD9, CD63, and CD81, which are widely used as canonical surface markers.
- Microvesicles (100 nm–1 µm): Shed directly from the plasma membrane through outward budding. Their surface protein content closely mirrors that of the parent cell membrane.
- Apoptotic bodies (1–5 µm): Released during programmed cell death. While less studied for intercellular signaling, they carry phosphatidylserine and damage-associated molecular patterns on their surface.
The surface protein landscape varies considerably across these subtypes and is further shaped by the cell of origin, the physiological state of that cell, and the microenvironmental stimuli it experiences. This context-dependence is precisely what makes EV surface proteomics such a powerful tool for monitoring cellular states in real time.
The Surface Proteome: A Molecular Identity Card
The proteins displayed on the outer leaflet of an extracellular vesicle serve multiple critical functions. They mediate target cell recognition, facilitate uptake, modulate immune responses, and encode information about the cell of origin. The EV surface proteome can be conceptually organized into several functional categories:
Tetraspanins and Scaffold Proteins
Tetraspanins — including CD9, CD63, CD81, and CD151 — are the most extensively characterized EV surface proteins. These four-transmembrane proteins organize membrane microdomains known as tetraspanin-enriched microdomains (TEMs). They interact with integrins, growth factor receptors, and immunoglobulin superfamily members, acting as molecular organizers that concentrate specific protein complexes on the EV surface.
Research from Théry et al. (2018) established consensus guidelines for EV characterization and highlighted tetraspanins as essential reference markers. Beyond their marker utility, tetraspanins actively shape EV tropism: CD63 promotes lysosomal targeting in recipient cells, while CD81 has been implicated in hepatocyte entry of hepatitis C virus — a finding with significant implications for EV-mediated pathogen dissemination (Boucheix & Rubinstein, 2001).
Integrins and Adhesion Molecules
Integrins on the EV surface are perhaps the best-characterized determinants of organ-specific homing. A landmark study by Hoshino et al. (2015), published in Nature, demonstrated that tumor-derived exosome integrins direct metastatic organotropism. Exosomes expressing integrin α6β4 preferentially fused with lung-resident cells, while those carrying integrin αvβ5 were taken up by liver Kupffer cells. This organ-specific targeting — encoded in surface proteins — was shown to prime pre-metastatic niches before the arrival of tumor cells, fundamentally reshaping our understanding of cancer metastasis.
Cell adhesion molecules (CAMs) such as ICAM-1, VCAM-1, and E-selectin are also found on EV surfaces, particularly on vesicles derived from endothelial cells under inflammatory conditions. These molecules facilitate EV docking to activated leukocytes, positioning EVs as active participants in immune cell recruitment.
Receptor Tyrosine Kinases and Growth Factor Receptors
Extracellular vesicles can carry functional growth factor receptors on their surfaces. EGFR (epidermal growth factor receptor), HER2, and MET have all been detected on tumor-derived EVs. Mechanistically, receptor-bearing EVs can transfer signaling competence to recipient cells — a phenomenon termed ‘receptor donation.’ This has profound implications: tumor cells may use EV-mediated receptor transfer to confer drug resistance or proliferative advantage to neighboring cells.
Studies in glioblastoma have shown that EGFR-amplified tumor cells shed EVs laden with mutant EGFRvIII. When these EVs are internalized by receptor-negative cells, they transiently activate downstream MAPK and Akt pathways, promoting recipient cell survival (Al-Nedawi et al., 2008). This mechanism underscores how EVs can effectively extend the phenotypic influence of a cell well beyond its immediate environment.
Immune Modulatory Proteins
The immune landscape of EV surfaces is remarkably complex. EVs from antigen-presenting cells (APCs) display MHC-I and MHC-II molecules along with co-stimulatory ligands such as CD80 and CD86, enabling them to directly stimulate or tolerize T cells without requiring cell-cell contact. This has earned EVs the designation of ‘long-range immune messengers.’
Conversely, tumor-derived EVs frequently display immunosuppressive surface proteins including PD-L1, which engages PD-1 on T cells to dampen anti-tumor immunity. Checkpoint inhibitor resistance in several cancer types has been mechanistically linked to PD-L1-enriched circulating exosomes (Chen et al., 2018). Targeting this surface interaction represents a growing area of therapeutic investigation.
How EV Surface Proteins Mediate Cellular Communication
The communication cycle mediated by EV surface proteins can be broken down into discrete molecular steps:
Step 1 — Biogenesis and Protein Sorting
Surface protein loading onto EVs is not a passive, stochastic process. Specific sorting signals direct proteins toward nascent vesicles. For endosome-derived exosomes, ESCRT (endosomal sorting complexes required for transport) machinery recognizes ubiquitinated cargo, while tetraspanin-enriched microdomains concentrate select transmembrane proteins independently of ubiquitination. Lipid composition, particularly ceramide and cholesterol content, further shapes the surface proteome by creating membrane curvature that favors incorporation of certain proteins.
Step 2 — Target Cell Recognition
Once secreted, EVs navigate biofluids and encounter potential recipient cells. Surface protein-receptor interactions on target cells determine whether an EV is internalized, ignored, or passed on. Lectins and heparan sulfate proteoglycans on target cell surfaces act as non-specific docking sites, while specific integrin-ligand, receptor-ligand, and antibody-antigen-like interactions provide selectivity. The net result is a remarkably sophisticated targeting system achieved without the machinery of adaptive immunity.
Step 3 — Uptake and Signal Delivery
EV uptake occurs via multiple routes: clathrin-mediated endocytosis, macropinocytosis, caveolin-dependent pathways, and direct membrane fusion. The choice of entry route is, in part, determined by surface proteins. For example, phosphatidylserine-binding receptors such as TIM-4 and stabilin-2 promote macropinocytic uptake of apoptotic EVs, while specific integrin-ligand interactions favor clathrin-mediated entry.
Once inside, EVs may fuse with endosomal membranes to release luminal cargo, or their surface proteins may continue to signal within the endolysosomal network. The fate of delivered surface receptors — recycling, degradation, or re-routing to the plasma membrane — remains an active area of investigation.
EV Surface Proteins as Biomarkers: Clinical Implications
The surface proteome of circulating EVs reflects the molecular phenotype of the cells that produced them. This has positioned EV surface protein profiling as one of the most promising frontiers in liquid biopsy — the non-invasive sampling of disease biomarkers from blood, urine, or other biofluids.
Cancer Diagnostics
In oncology, tumor-derived EVs carrying tumor-specific surface antigens can be detected in plasma months before conventional imaging reveals a lesion. EpCAM (epithelial cell adhesion molecule), EGFR, HER2, and PSA have all been detected on circulating EV surfaces in cancer patients. High-sensitivity platforms including nano-plasmonic sensing, single-vesicle flow cytometry, and the nanoparticle-based ExoChip array have demonstrated the ability to detect sub-picomolar concentrations of antigen-bearing EVs.
A pivotal study by Melo et al. (2015) identified glypican-1 (GPC1) as a surface marker specifically enriched on pancreatic cancer-derived exosomes, with 100% sensitivity and specificity in distinguishing cancer patients from healthy controls in the reported cohort. While subsequent studies have moderated this initial optimism by identifying biological variability, GPC1-positive EVs remain among the most rigorously investigated EV biomarker candidates in solid tumors.
Neurological Disorders
The blood-brain barrier presents a formidable obstacle to conventional CNS biomarker sampling. However, brain-derived EVs cross this barrier into systemic circulation, carrying surface proteins characteristic of their neural origin. L1CAM (CD171), a neuronal cell adhesion molecule, has been used to immunocapture neuronally-derived EVs from plasma in studies of Alzheimer’s disease, traumatic brain injury, and Parkinson’s disease. Surface protein analysis of L1CAM-positive EVs has revealed elevated amyloid-β, phosphorylated tau, and α-synuclein levels, opening new avenues for early CNS disease detection.
Cardiovascular Disease
Cardiomyocyte- and endothelial-derived EVs bearing surface proteins such as cardiac troponin, PECAM-1 (CD31), and von Willebrand factor have been detected in elevated concentrations following myocardial injury. Surface proteomic profiling of EVs from heart failure patients has identified distinct molecular signatures that correlate with left ventricular ejection fraction, offering potential monitoring tools that complement or surpass conventional serum markers.
Engineering EV Surface Proteins for Therapeutic Applications
The precise control of EV surface composition presents an unparalleled opportunity for therapeutic engineering. By modifying surface proteins, researchers can create EVs with enhanced targeting specificity, improved immune evasion, and programmable signaling activity.
Targeted Drug Delivery
Genetic and chemical strategies have been employed to display targeting ligands on EV surfaces. Fusion proteins combining the EV-enriched protein LAMP2B with organ-specific peptides (e.g., the neuron-targeting RVG peptide) enable EVs to selectively deliver siRNA payloads to the central nervous system following systemic administration (Alvarez-Erviti et al., 2011). Similarly, anti-HER2 nanobodies anchored to the EV surface via GPI-anchor sequences have shown selective uptake by HER2-positive breast cancer cells in vitro and in vivo.
Immune Modulation
Engineered EVs displaying tolerogenic surface proteins — such as PD-L1, CTLA-4-Ig fusion proteins, or antigen-MHC complexes — are being explored as antigen-specific immunosuppressive agents for autoimmune disease and transplant tolerance. Conversely, EVs engineered to present pro-inflammatory cytokine anchors or tumor-associated antigens are in early-phase investigation as cancer vaccines.
Surface Protein Display Platforms at LuminEV
At LuminEV, our proprietary surface engineering platforms enable precise control over EV surface protein composition. Using a combination of genetic engineering, post-secretion click chemistry functionalization, and high-throughput proteomics validation, we produce EVs with defined surface identities suitable for research, diagnostic tool development, and preclinical therapeutic evaluation. Our nanoproteomic characterization pipeline provides single-vesicle resolution surface protein profiling, moving beyond population-average measurements that obscure the true diversity of EV surface landscapes.
Analytical Technologies for EV Surface Protein Profiling
Accurate characterization of EV surface proteins demands specialized analytical approaches that account for the nanoscale dimensions of vesicles and the low abundance of specific surface epitopes.
- Nano-flow cytometry: Next-generation instruments such as the NanoFCM platform can resolve individual vesicles as small as 40 nm and simultaneously measure forward scatter (size) and fluorescence intensity from surface-labeled antibodies, enabling multiplexed single-vesicle phenotyping.
- Proximity ligation assay (PLA): Paired antibody probes linked to oligonucleotide arms generate amplifiable ligation products only when both target proteins are present on the same vesicle surface, enabling ultrasensitive co-expression analysis.
- Mass spectrometry-based surface proteomics: Surface protein-selective biotinylation followed by streptavidin enrichment and LC-MS/MS provides comprehensive, quantitative surface proteome maps with sub-femtomole sensitivity.
- Surface plasmon resonance (SPR) and localized SPR (LSPR): Label-free optical biosensors capable of real-time monitoring of EV-receptor binding kinetics, providing affinity and rate constants for EV surface protein interactions.
- Cryo-electron tomography: Emerging structural techniques now allow 3D reconstruction of EV surface architecture at nanometer resolution, revealing the spatial organization of surface proteins in their native lipid bilayer context.
Challenges and Future Directions
Despite rapid advances, several challenges must be addressed to fully harness the potential of EV surface proteomics:
Heterogeneity and standardization: EV populations are inherently heterogeneous, and the lack of universal isolation and characterization standards has complicated cross-study comparisons. The MISEV2023 guidelines from the International Society for Extracellular Vesicles (ISEV) represent the latest consensus effort to standardize EV research practices.
Single-vesicle resolution: Population-average proteomic methods mask the true diversity of surface protein co-expression across individual vesicles. Next-generation single-vesicle platforms that combine multi-parameter phenotyping with functional readouts will be essential for understanding EV subpopulation biology.
In vivo fate tracking: Despite elegant in vitro binding studies, the actual in vivo biodistribution of specific EV surface protein phenotypes remains poorly characterized. Non-invasive EV tracking technologies — including radiolabeling, bioluminescence reporters anchored to surface proteins, and photoacoustic imaging — are under active development.
Translation to clinical diagnostics: Converting laboratory EV surface biomarker discoveries into validated clinical tests requires large prospective cohort validation, assay standardization, and regulatory clearance pathways that the field is only beginning to navigate.
Conclusion
Extracellular vesicle surface proteins are not merely structural features — they are the molecular syntax of intercellular communication. They encode the identity of the cell of origin, specify the recipient cells an EV will engage, and orchestrate the downstream functional responses that shape tissue physiology and disease progression.
The convergence of advances in single-vesicle analytics, surface engineering, and clinical biomarker validation is rapidly transforming EV surface proteomics from a specialized research tool into a clinically actionable discipline. From cancer liquid biopsies and neurological disease monitoring to precision-targeted drug delivery systems, the ability to read and write the language of EV surface proteins stands at the forefront of next-generation medicine.
At LuminEV, we are committed to advancing the science and application of extracellular vesicle surface proteomics. Our integrated platform — spanning EV production, surface engineering, and analytical characterization — is designed to accelerate discovery and translate insights into real-world impact. We invite researchers, clinicians, and industry partners to explore how EV surface protein science can inform and advance their work.
References
Al-Nedawi K, Meehan B, Micallef J, et al. (2008). Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nature Cell Biology, 10(5), 619–624. https://doi.org/10.1038/ncb1725
Alvarez-Erviti L, Seow Y, Yin H, et al. (2011). Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nature Biotechnology, 29(4), 341–345. https://doi.org/10.1038/nbt.1807
Boucheix C, Rubinstein E. (2001). Tetraspanins. Cellular and Molecular Life Sciences, 58(9), 1189–1205. https://doi.org/10.1007/PL00000933
Chen G, Huang AC, Zhang W, et al. (2018). Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature, 560(7718), 382–386. https://doi.org/10.1038/s41586-018-0392-8
Hoshino A, Costa-Silva B, Shen TL, et al. (2015). Tumour exosome integrins determine organotropic metastasis. Nature, 527(7578), 329–335. https://doi.org/10.1038/nature15756
Lötvall J, Hill AF, Hochberg F, et al. (2014). Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. Journal of Extracellular Vesicles, 3(1), 26913. https://doi.org/10.3402/jev.v3.26913
Melo SA, Luecke LB, Kahlert C, et al. (2015). Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature, 523(7559), 177–182. https://doi.org/10.1038/nature14581
Théry C, Witwer KW, Aikawa E, et al. (2018). Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles. Journal of Extracellular Vesicles, 7(1), 1535750. https://doi.org/10.1080/20013078.2018.1535750
van Niel G, D’Angelo G, Raposo G. (2018). Shedding light on the cell biology of extracellular vesicles. Nature Reviews Molecular Cell Biology, 19(4), 213–228. https://doi.org/10.1038/nrm.2017.125
Welsh JA, Goberdhan DCI, O’Driscoll L, et al. (2024). Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. Journal of Extracellular Vesicles, 13(2), e12404. https://doi.org/10.1002/jev2.12404